QuantiChrom™ Chloride Assay Kit
Application
- For quantitative determination of chloride ion and evaluation of drug effects on chloride metabolism.
Key Features
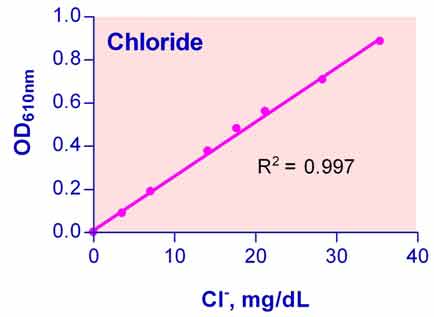
- Sensitive and accurate. Use as little as 5 µL samples. Linear detection range 0.7 mg/dL (0.2 mM) to 35 mg/dL (10 mM) Cl– in 96-well plate assay.
- Simple and high-throughput. The procedure involves the addition of a single working reagent and incubation for 5 min. Can be readily automated as a high-throughput assay for thousands of samples per day.
- Improved reagent stability and versatility. The optimized formulation has greatly enhanced the reagent and signal stability. Cuvet or 96-well plate assay.
- Low interference in biological samples. No pretreatments are needed. Assays can be directly performed on raw biological samples i.e., in the presence of lipids, proteins, and minerals such as magnesium, iron, and zinc.
Method
- OD610nm
Samples
- Biological, food, and environment
Species
- All
Procedure
- 5 min
Size
- 250 tests
Detection Limit
- 0.7 mg/dL (0.2 mM)
Shelf Life
- 12 months
More Details
Chloride is the major extracellular anion in human body fluids. Chloride plays a key role in maintaining proper water distribution, osmotic pressure, and electrolyte balance. Low chloride concentrations may be found with prolonged vomiting, extensive burns, metabolic acidosis, Addisonia crisis, and renal diseases. Elevated chloride concentrations are associated with dehydration, congestive heart failure, hyperventilation and urinary obstructions. Determination of chloride in sweat is useful in diagnosing cystic fibrosis. Simple, direct, and automation-ready procedures for measuring chloride concentration in biological samples are becoming popular in Research and Drug Discovery. BioAssay Systems chloride assay kit is designed to measure chloride directly in biological samples without any pretreatment. The improved Fried method utilizes mercuric 2,4,6-tripyridyl-s-triazine, which forms a colored complex specifically with chloride. The intensity of the color, measured at 610nm, is directly proportional to the chloride concentration in the sample. The optimized formulation substantially reduces interference by substances in the raw samples.No frequently asked questions for this new product. Please check back later.
For more detailed product information and questions, please feel free to email us. Or for more general information regarding our assays, please refer to Technical Support.
Daniel, J et al (2020). Total-tract digestibility and milk productivity of dairy cows as affected by trace mineral sources. Journal of Dairy Science, 103(10), 9081-9089. Assay: Chloride in mash compound feed and forages.
Prinsi, B et al (2020). Root proteomic analysis of two grapevine rootstock genotypes showing different susceptibility to salt stress. International Journal of Molecular Sciences, 21(3), 1076. Assay: Chloride in grapevine root system.
Kim, H. Y., Rhyu, M. R., Kim, K., Park, M., & Yu-Mi, L. E. E. (2019). U.S. Patent Application No. 10/293,009. Assay: Chloride in rats blood.
Wilms, J., Berends, H., & Martin-Tereso, J. (2019). Hypertonic milk replacers increase gastrointestinal permeability in healthy dairy calves. Journal of dairy science, 102(2), 1237-1246. Assay: Chloride in calvse milk replacer.
Wilms, J., Wang, G., Doelman, J., Jacobs, M., & Martin-Tereso, J. (2019). Intravenous calcium infusion in a calving protocol disrupts calcium homeostasis compared with an oral calcium supplement. Journal of dairy science 102(7):6056-6064. Assay: Chloride in feed.
Lin, W., Das, K., Degen, D., Mazumder, A., Duchi, D., Wang, D. & Srivastava, A. (2018). Structural basis of transcription inhibition by fidaxomicin (lipiarmycin A3). Molecular cell, 70(1), 60-71. Assay: Chloride in E. coli.
Tanaka, S., Miyazaki, H., Shiozaki, A., Ichikawa, D., Otsuji, E., & Marunaka, Y. (2017). Cytosolic Cl-affects the anticancer activity of paclitaxel in the gastric cancer cell line, MKN28 Cell. Cellular Physiology and Biochemistry, 42(1), 68-80. Assay: Chloride in human gastric adenocarcinoma mkn28.
Hannan, K. D. (2016). The physiological effects of elevated carbon dioxide, in the context of non-physical fish barriers, on unionid mussels (Doctoral dissertation). Assay: Chloride in Fusconaia flava hemolymph.
Ballester-Lozano GF et al. (2015). Comprehensive biometric, biochemical and histopathological assessment of nutrient deficiencies in gilthead sea bream fed semi-purified diets. Br J Nutr. 114(5):713-26. Assay: chloride in fish plasma.
Everaert N, et al (2011). Regulatory capacities of a broiler and layer strain exposed to high CO2 levels during the second half of incubation. Comp Biochem Physiol A Mol Integr Physiol. 158(2):215-20. Assay: Chloride in chicken plasma.
Grunberg W, et al (2011). Periparturient effects of feeding a low dietary cation-anion difference diet on acid-base, calcium, and phosphorus homeostasis and on intravenous glucose tolerance test in high-producing dairy cows. J Dairy Sci. 94(2):727-45. Assay: Chloride in cow urine.
Peng S,et al (2010). Minimal effects of VEGF and anti-VEGF drugs on the permeability or selectivity of RPE tight junctions. Invest Ophthalmol Vis Sci. 51(6):3216-25. Assay: Chloride in human junction liquid.
Rajapakshe A, et al (2010). Characterization of chloride-depleted human sulfite oxidase by electron paramagnetic resonance spectroscopy: experimental evidence for the role of anions in product release. Biochemistry49(25):5154-9. Assay: Chloride in human sulfite oxidase.
Ahmad T, et al (2009). Influence of varying dietary electrolyte balance on broiler performance under tropical summer conditions. J Anim Physiol Anim Nutr (Berl). 93(5):613-21. Assay: Chloride in broiler chick blood.
Borenshtein D, et al (2009). Decreased expression of colonic Slc26a3 and carbonic anhydrase iv as a cause of fatal infectious diarrhea in mice. Infect Immun. 77(9):3639-50. Assay: Chloride in mice serum.
Jensen SB, et al (2008). Adjuvant chemotherapy in breast cancer patients induces temporary salivary gland hypofunction. Oral Oncol. 44(2):162-73. Assay: Chloride in human serum.
To find more recent publications, please click here.
If you or your labs do not have the equipment or scientists necessary to run this assay, BioAssay Systems can perform the service for you.
– Fast turnaround
– Quality data
– Low cost
Please email or call 1-510-782-9988 x 2 to discuss your projects.
$399.00
For bulk quote or custom reagents, please email or call 1-510-782-9988 x 1.
Orders are shipped the same day if placed by 2pm PST
Shipping: RT
Carrier: Fedex
Delivery: 1-2 days (US), 3-6 days (Intl)
Storage: 4°C upon receipt
Related Products
You may also like…
| Name | SKU | Price | Buy |
|---|---|---|---|
| QuantiChrom™ Copper Assay Kit | DICU-250 |
$549.00 |
|
| QuantiChrom™ Iron Assay Kit | DIFE-250 |
$499.00 |
|
| QuantiChrom™ Magnesium Assay Kit | DIMG-250 |
$489.00 |
|
| QuantiChrom™ Calcium Assay Kit | DICA-500 |
$459.00 |
|
| QuantiChrom™ Chromium Assay Kit | DCRM-250 |
$519.00 |
Why BioAssay Systems
Quality and User-friendly • Expert Technical Support • Competitive Prices • Expansive Catalogue • Trusted Globally
