Malachite Green Phosphate Assay Kit
Application
- For sensitive and high-throughput phosphate determination.
Key Features
- Reagent is very stable. Due to our innovative formulation, no precipitation of reagent occurs. Therefore no filtration of reagent is needed prior to assays, as is often required with other commercial kits.
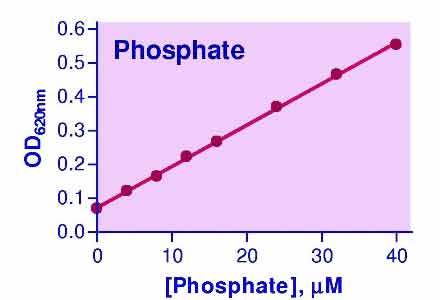
- High sensitivity and wide detection range: detection of as little of 1.6 pmoles of phosphate and useful range between 0.02 µM and 40 µM phosphate.
- Fast and convenient: homogeneous “mix-and-measure” assay allows quantitation of free phosphate within 20 minutes.
- Compatible with routine laboratory and HTS formats: assays can be performed in tubes, cuvettes or microplates, on spectrophotometers and plate readers. Robust and amenable to HTS: Z factors of 0.7 to 0.9 are observed in 96-well and 384-well plates. Can be readily automated on HTS liquid handling systems.
Method
- OD620nm (malachite green)
Samples
- Biological, environment etc
Species
- All
Procedure
- 30 min
Size
- 2500 tests
Detection Limit
- 0.02 µM
Shelf Life
- 12 months
More Details
The Malachite Green Phosphate Assay Kit is based on the quantification of the green complex formed between Malachite Green, molybdate and free orthophosphate. The rapid color formation from the reaction can be conveniently measured on a spectrophotometer (600 – 660 nm) or on a plate reader. The non-radioactive colorimetric assay kits have been optimized to offer superior sensitivity and prolonged shelf life. The assay is simple and fast, involving a single addition step for phosphate determination. Assays can be executed in tubes, cuvettes or multi-well plates. The assays can be conveniently performed in 96- and 384-well plates for high-throughput screening of enzyme inhibitors.What is the difference between your 3 phosphate assays: DIPI-500, POMG-25H, POPB-500?
All three products are based on the malachite green dye. While the PiBlue (POPB-500) and QuantiChrom (DIPI-500) Phosphate Assay Reagents are identical, they differ in the standards. The PiBlue is designed for customers who want prepare a full calibration curve for their assays, and the latter has one set of a blank control and 30 µM phosphate standard. The reagent is more diluted than the reagent used in the Malachite Green Phosphate Assay Kit (POMG-25H). As a result, one adds 100 µL reagent (POPB-500 or DIPI-500) to 50 µL sample, whereas for the Malachite Green based assay, one adds 20 µL Reagent to 80 µL sample.
In practice, the DIPI-500 is the most convenient kit to use, because there is no need to prepare a calibration curve there is no need for extra mixing. Because adding a larger volume (100 μL) to a small volume (50 μL) effects efficient mixing in well, no extra mixing is needed. The POPB-500 requires preparation of calibration curve. With the POMG-25H Kit, one needs to mix the reagent with the sample, because adding 20 µL to 80 µL does not mix efficiently by itself. Also a preparation of a standard curve is necessary.
I’m using the Malachite Green Phosphate Assay Kit (POMG-25H) to detect GTPase activity. However, I saw high background and no increase in phosphate liberation.
This assay is very sensitive to free phosphate. Before each assay, it is important to check that all materials (substrates, enzyme preparations and buffers) are pure and free of contaminating phosphate. This can be conveniently done by adding 20 µL of the Working Reagent to 80 µL sample solution. The blank OD values at 620 nm should be lower than 0.2. If the OD readings are higher than 0.2, check water phosphate level. Double distilled water usually have OD readings lower than 0.1. Lab detergents may contain high levels of phosphate. Make sure that lab wares are free from contaminating phosphate after thorough washes.
In the case of enzyme reactions, the reaction may be terminated directly by the addition of the acidic Working Reagent. Dilution of reaction mixture may be necessary prior to the assay. For ATPase or GTPase assays, the ATP or GTP concentration should be lower than 0.25 mM. If the reaction mixture contains > 0.25mM ATP or GTP, dilute samples in distilled water. For example, if the ATPase reaction contained 1 mM ATP, at the end of reaction dilute reaction mixture 4-fold in water prior to the assay.
The chromogenic reaction is completed within 30 min at room temperature. Read OD at or shorter than 30 min to minimize background due to slow hydrolysis of the substrates (ATP or GTP) by the acidic reagent.
If the background OD is in the normal range, but there is still no free phosphate detected, it is likely the GTPase was not active. To confirm this possibility, one could run a test without the enzyme by spiking free phosphate (e.g. 0.5 – 40 µM) into the mixture of Buffer and Substrate. In this case, one should see an increase in OD with increasing free phosphate concentrations. This confirms that the enzyme was not active in the assay.
Zhao, JY et al (2020). Atherogenic diet accelerates ectopic mineralization in a mouse model of pseudoxanthoma elasticum. International Journal of Dermatology and Venereology, 3(2), 91-96. Assay: Phosphate in serum mouse.
Lee, JS et al (2020). Host surface ectonucleotidase-CD73 and the opportunistic pathogen, Porphyromonas gingivalis, cross-modulation underlies a new homeostatic mechanism for chronic bacterial survival in human epithelial cells. Virulence, 11(1), 414-429. Assay: Phosphate in human epithelial cells.
Chauhan, AS et al (2019). Trafficking of a multifunctional protein by endosomal microautophagy: Linking two independent unconventional secretory pathways. FASEB Journal: Official Publication of the Federation of American Societies for Experimental Biology, 33(4), 5626-5640. Assay: Phosphate in mouse cell culture supernatant.
Dangi, P et al (2019). Natural product inspired novel indole based chiral scaffold kills human malaria parasites via ionic imbalance mediated cell death. Scientific Reports, 9(1), 17785. Assay: Phosphate in plasmodium falciparum membrane fraction.
Wang, X et al (2020). The antitumor agent ansamitocin p-3 binds to cell division protein ftsz in actinosynnema pretiosum. Biomolecules, 10(5). Assay: Phosphate in actinosynnema pretiosum.
Ju, S et al (2020). Oxygenated polycyclic aromatic hydrocarbons from ambient particulate matter induce electrophysiological instability in cardiomyocytes. Particle and Fibre Toxicology, 17(1), 25. Assay: Phosphate in rat cardiomyocyte microsome membranes.
Updegrove, TB et al (2021). Reformulation of an extant ATPase active site to mimic ancestral GTPase activity reveals a nucleotide base requirement for function. ELife, 10. Assay: Phosphate in bacillus subtilis GTPase reaction mixture.
Wang, J et al (2020). Tumor-responsive, multifunctional CAR-NK cells cooperate with impaired autophagy to infiltrate and target glioblastoma. BioRxiv, 2020.10.07.330043. Assay: Phosphate in mouse cells.
Afshar, N et al (2021). A novel motif of Rad51 serves as an interaction hub for recombination auxiliary factors. ELife, 10. Assay: Phosphate in schizosaccharomyces pombe proteins.
Ito, K et al (2020). Real-time tracking reveals catalytic roles for the two DNA binding sites of Rad51. Nature Communications, 11(1), 2950. Assay: Phosphate in schizosaccharomyces pombe proteins.
Moore, M., Moriarty, T. A., Connolly, G., Mermier, C., Amorim, F., Miller, K., & Zuhl, M. (2019). Oral Glutamine Supplement Reduces Subjective Fatigue ratings during Repeated Bouts of Firefighting Simulations. Safety 5(2), 38. Assay: Antioxidant in human serum.
Soni, V et al (2015). Depletion of M. tuberculosis GlmU from Infected Murine Lungs Effects the Clearance of the Pathogen. PLoS Pathog 11(10):e1005235. Assay: Phosphate in bacteria enzyme.
Taib, Izatus Shima, et al.(2015) “Palm oil tocotrienol-rich fraction attenuates testicular toxicity induced by fenitrothion via an oxidative stress mechanism.” Toxicology Research 4.1: 132-142. Assay: Sialic Acid in rat testis.
Aldana-Masangkay GI,et al (2011). Tubacin suppresses proliferation and induces apoptosis of acute lymphoblastic leukemia cells. Leuk Lymphoma. 52(8):1544-55. Assay: Phosphate in human cell.
Cha C, et al (2011). Integrative design of a poly(ethylene glycol)-poly(propylene glycol)-alginate hydrogel to control three dimensional biomineralization. Biomaterials.32(11):2695-703. Assay: Phosphate in hydrogels.
Demidenko AA, et al (2011). Effects of viscogens on RNA transcription inside reovirus particles. J Biol Chem. 286(34):29521-30. Assay: Phosphate in mouse enzyme extract.
Khuda-Buksh, AR. et al. (2011). Analysis of the capability fo ultra-highly diluted glucose to increase glucose uptake in arsenite-streesed bacteria Escherichia coli. J, Chin. Integrative Medicine 9(8): 901-912. Assay: phosphate in bacteria cell lysate.
Lin J, Wilson MA (2011). Escherichia coli thioredoxin-like protein YbbN contains an atypical tetratricopeptide repeat motif and is a negative regulator of GroEL. J. Biol Chem. 286(22):19459-69. Assay: Phosphate in bacteria enzyme.
Lin J, Wilson MA (2011). Escherichia coli thioredoxin-like protein YbbN contains an atypical tetratricopeptide repeat motif and is a negative regulator of GroEL. J. Biol Chem. 286(22):19459-69. Assay: Phosphate in plant extracts.
Liu et al (2011). Competition between foliar Neotyphodium lolii endophytes and mycorrhizal Glomus spp. fungi in Lolium perenne depends on resource supply and host carbohydrate content. Functional Ecology 25:910-920. Assay: Phosphate in plant extracts.
Reigan P, et al (2011). A mechanistic and structural analysis of the inhibition of the 90-kDa heat shock protein by the benzoquinone and hydroquinone ansamycins. Mol Pharmacol. 79(5):823-32. Assay: Phosphate in human Hsp90.
Hsieh CW, et al (2010). Direct MinE-membrane interaction contributes to the proper localization of MinDE in E. coli. Mol Microbiol. 75(2):499-512. Assay: Phosphate in bacteria ATPase, GTPase.
Hu, M et al. (2010) Effect of prolonged starvation on body weight and blood-chemistry in two horseshoe crab species: Tachypleus tridentatus and Carcinoscorpius rotundicauda (Chelicerata: Xiphosura). J. Exp Marine Biol Ecology 395(1-2):112-119. Assay: Cholesterol in crab plasma.
Li Q, Uitto J (2010). The mineralization phenotype in Abcc6 ( -/- ) mice is affected by Ggcx gene deficiency and genetic background–a model for pseudoxanthoma elasticum. J Mol Med (Berl). 88(2):173-81. Assay: Phosphate in mouse tissue.
Yoshida C et al (2010). Analysis of inhibition of topoisomerase IIalpha and cancer cell proliferation by ingenolEZ. Cancer Sci. 101(2):374-8. Assay: Phosphate in bacteria DNA hydrolysis (S. thermophilus).
Yoshida C, et al (2010). Analysis of inhibition of topoisomerase IIalpha and cancer cell proliferation by ingenolEZ. Cancer Sci. 101(2):374-8. Assay: Phosphate in human topoisomerase.
Barisic S, et al (2008). Identification of PP2A as a crucial regulator of the NF-kappaB feedback loop: its inhibition by UVB turns NF-kappaB into a pro-apoptotic factor. Cell Death Differ. 15(11):1681-90. Assay: Phosphate in human cell lysate, PP2A.
Adkins, M.W. et al (2007). Chromatin Disassembly from the PHO5 Promoter Is Essential for the Recruitment of the General Transcription Machinery and Coactivators. Mol. Cell. Biol. 27: 6372-6382. Assay: Phosphate in yeast culture.
Anand SP, et al (2007). DNA helicase activity of PcrA is not required for the displacement of RecA protein from DNA or inhibition of RecA-mediated strand exchange. J Bacteriol. 189(12):4502-9. Assay: Phosphate in bateria enzyme.
Guerette D et al (2007). Molecular evolution of type VI intermediate filament proteins. BMC Evolutionary Biology 7:164. Assay: Phosphate in bacteria GTP hydrolysis.
Guerette, D. et al (2007). Molecular evolution of type VI intermediate filament proteins. BMC Evolutionary Biology 7:164. Assay: Phosphate in human ATPase, GTPase.
Koroleva O, et al (2007). Structural conservation of RecF and Rad50: implications for DNA recognition and RecF function. EMBO J. 26(3):867-77. Assay: Phosphate in bacteria enzyme.
Lu B, et al (2007). Roles for the human ATP-dependent Lon protease in mitochondrial DNA maintenance. J Biol Chem. 282(24):17363-74. Assay: Phosphate in human enzyme.
Blumental-Perry A, et al (2006). Phosphatidylinositol 4-phosphate formation at ER exit sites regulates ER export. Dev Cell. 11(5):671-82. Assay: Phosphate in rat liposomes.
Guo W, et al (2006). The bioreduction of a series of benzoquinone ansamycins by NAD(P)H:quinone oxidoreductase 1 to more potent heat shock protein 90 inhibitors, the hydroquinone ansamycins. Mol Pharmacol. 70(4):1194-203. Assay: Phosphate in yeast purified ATPase.
Saran, D et al (2006). Multiple-turnover thio-ATP hydrolase and phospho-enzyme intermediate formation activities catalyzed by an RNA enzyme. Nucleic Acids Res. 34(11): 3201-3208. Assay: Phosphate in human phosphotyrosyl phosphatase activator.
Saran, D. et al (2006). Multiple-turnover thio-ATP hydrolase and phospho-enzyme intermediate formation activities catalyzed by an RNA enzyme. Nucleic Acids Res. 34(11): 3201-3208. Assay: Phosphate in human RNA.
Green, M.L. et al (2005). Ethylene glycol induces hyperoxaluria without metabolic acidosis in rats. Am J Physiol Renal Physiol 289: F536-F543. Assay: Phosphate in rat urine, serum.
Guo W, et al (2005). Formation of 17-allylamino-demethoxygeldanamycin (17-AAG) hydroquinone by NAD(P)H:quinone oxidoreductase 1: role of 17-AAG hydroquinone in heat shock protein 90 inhibition. Cancer Res. 65(21):10006-15. Assay: Phosphate in human Hsp90.
Kim HE,et al (2005).Formation of apoptosome is initiated by cytochrome c-induced dATP hydrolysis and subsequent nucleotide exchange on Apaf-1. PNAS 102(49):17545-50. Assay: Phosphate in human enzyme.
To find more recent publications, please click here.
If you or your labs do not have the equipment or scientists necessary to run this assay, BioAssay Systems can perform the service for you.
– Fast turnaround
– Quality data
– Low cost
Please email or call 1-510-782-9988 x 2 to discuss your projects.
$329.00
For bulk quote or custom reagents, please email or call 1-510-782-9988 x 1.
Orders are shipped the same day if placed by 2pm PST
Shipping: RT
Carrier: Fedex
Delivery: 1-2 days (US), 3-6 days (Intl)
Storage: 4°C upon receipt
Related Products
You may also like…
| Name | SKU | Price | Buy |
|---|---|---|---|
| PiBlue™ Phosphate Assay Kit | POPB-500 |
$279.00 |
Why BioAssay Systems
Quality and User-friendly • Expert Technical Support • Competitive Prices • Expansive Catalogue • Trusted Globally
