EnzyChrom™ Glycogen Assay Kit
Application
- For quantitative determination of glycogen and evaluation of drug effects on glycogen metabolism.
Key Features
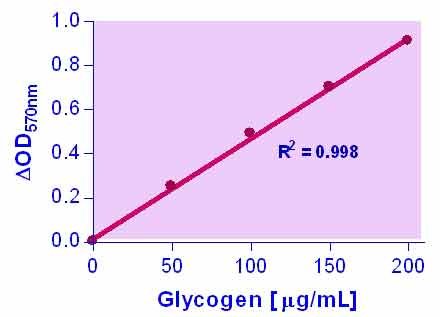
- Use as little as 10 µL samples. Linear detection range: 2 to 200 µg/mL glycogen for colorimetric assays and 0.2 to 20 µg/mL for fluorimetric assays.
Method
- OD570nm, or FL530/585nm
Samples
- Biological
Species
- All
Procedure
- 30 min
Size
- 100 tests
Detection Limit
- OD, FL: 2, 0.2 µg/mL
Shelf Life
- 6 months
More Details
GLYCOGEN is a branched polysaccharide of glucose units linked by α-1,4 glycosidic bonds and α-1,6 glycosidic bonds. It is stored primarily in the liver and muscle and forms an energy reserve that can be quickly mobilized to meet a sudden need for glucose. The most common glycogen metabolism disorder is found in diabetes, in which, due to abnormal amounts of insulin, liver glycogen can be abnormally accumulated or depleted. Genetic glycogen storage diseases have been associated with various inborn errors of metabolism caused by deficiencies of enzymes necessary for glycogen synthesis or breakdown. Simple, direct, and automation-ready procedures for measuring glycogen concentrations find wide applications in research and drug discovery. BioAssay Systems glycogen assay uses a single Working Reagent that combines the enzymatic breakdown of glycogen and the detection of glucose in one step. The color intensity of the reaction product at 570nm or fluorescence intensity at λex/em = 530/585 nm is directly proportional to the glycogen concentration in the sample. This simple convenient assay is carried out at room temperature and takes only 30 min.How to prepare cell samples for the glycogen assay?
We recommend a modified version of the protocol used by Murat & Serfaty (Clin Chem. 12:1576-7).
– Homogenize 10×6 cells with 200 µL buffer (0.025 M citrate, pH 4.2, 2.5 g/L NaF) on ice.
– Centrifuge sample at 14,000 x g for 5 min to remove debris, and use 10 µL of the clear supernatant in the assay.
– I would also recommend diluting the standard in this buffer rather than distilled water to increase the accuracy of the assay.
Tissue preparation: 10 to 30 mg/mL tissue in 0.025 M citrate, ph3.2, 2.5 g/L NaF.
2. I obtained a very high glycogen concentration after homogenizing mouse liver tissue in a sucrose/EDTA buffer. Indeed, the amount of glycogen was higher than the amount of liver tissue I added. A sample blank (without enzyme A) showed there is only a small amount of glucose in the sample. What could cause these results?
One of the enzymes in enzyme A, transglucosidase, hydrolyzes sucrose into glucose and fructose. You are actually measuring the amount of buffer rather than the amount of glycogen in your sample. We recommend using a sucrose free buffer, e.g. (0.025 M citrate, ph3.2, 5 g/L NaF).
Shi, J., et al (2020). Liver ChREBP protects against fructose-induced glycogenic Hepatotoxicity by regulating L-type pyruvate kinase. Diabetes, 69(4), 591-602. Assay: Glycogen in mouse liver tissue.
Price, C., et al (2020). Paradoxical pro-inflammatory responses by human macrophages to an amoebae host-adapted legionella effector. Cell Host & Microbe, 27(4), 571-584.e7. Assay: Glycogen in human macrophage cellls.
Liu, J., et al. (2020). CaMKIV limits metabolic damage through induction of hepatic autophagy by CREB in obese mice. Journal of Endocrinology, 244(2), 353-367. Assay: Glycogen in mouse serum.
Li, Y., et al (2021). Isolated plin5-deficient cardiomyocytes store less lipid droplets than normal, but without increased sensitivity to hypoxia. Biochimica et Biophysica Acta (BBA) – Molecular and Cell Biology of Lipids, 1866(4), 158873. Assay: Glycogen in mouse cardiomyocytes and heart tissue.
Lambert, K., et al. (2020). Biocompatible modified water as a non-pharmaceutical approach to prevent metabolic syndrome features in obesogenic diet-fed mice. Food and Chemical Toxicology, 141, 111403 Assay: Glycogen in mouse serum or liver tissue.
Hunter, A. L., et al (2020). Nuclear receptor REVERBalpha is a state-dependent regulator of liver energy metabolism. Proceedings of the National Academy of Sciences, 117(41), 25869-25879. Assay: Glycogen in mouse liver tissue.
Nunn, K. L., et al (2020). Amylases in the human vagina. mSphere, 5(6). Assay: Glycogen in cervicovaginal mucus.
Renquist, B. J., Madanayake, T. W., Ghimire, S., Geisler, C. E., Xu, Y., & Bogan, R. L. (2018). Transmembrane protein 135 (TMEM135) is a liver X receptor target gene that mediates an auxiliary peroxisome matrix protein import pathway. bioRxiv, 334979. Assay: Glycogen in mouse liver tissue.
Turkyilmaz, M. K., Dereli Fidan, E., Karaarslan, S., Unubol Aypak, S., & Nazligul, A. (2018). Effect of Preslaughter Shackling on Stress, Meat Qu ality Traits, and Glycolytic Potential in Broilers. Assay: Glycogen in broiler chicken muscle tissue.
Nakajima, T., Yoshikawa, K., Toya, Y., Matsuda, F., & Shimizu, H. (2017). Metabolic Flux Analysis of the Synechocystis sp. PCC 6803 Delta nrtABCD Mutant Reveals a Mechanism for Metabolic Adaptation to Nitrogen-Limited Conditions. Plant and Cell Physiology, 58(3), 537-545. Assay: Glycogen in Synechocystis cells.
Wang, X. X., Ye, T., Li, M., Li, X., Qiang, O., Tang, C. W., & Liu, R. (2017). Effects of octreotide on hepatic glycogenesis in rats with high fat diet-induced obesity. Molecular medicine reports, 16(1), 109-118. Assay: Glycogen in Sprague Dewley rats liver tissue.
Adeyemi, K. D., Shittu, R. M., Sabow, A. B., Abubakar, A. A., Karim, R., Karsani, S. A., & Sazili, A. Q. (2016). Comparison of myofibrillar protein degradation, antioxidant profile, fatty acids, metmyoglobin reducing activity, physicochemical properties and sensory attributes of gluteus medius and infraspinatus muscles in goats. Journal of animal science and technology, 58(1), 23. Assay: Glycogen in mice liver tissue.
Adeyemi, K. D., Shittu, R. M., Sabow, A. B., Ebrahimi, M., & Sazili, A. Q. (2016). Influence of diet and postmortem ageing on oxidative stability of lipids, myoglobin and myofibrillar proteins and quality attributes of gluteus medius muscle in goats. PloS one, 11(5), e0154603. Assay: Glycogen in goat muscle tissue.
Bastow, E. L., Peswani, A. R., Tarrant, D. S., Pentland, D. R., Chen, X., Morgan, A. & Tuite, M. F. (2016). New links between SOD1 and metabolic dysfunction from a yeast model of amyotrophic lateral sclerosis. J Cell Sci, 129(21), 4118-4129. Assay: Glycogen in yeast cells.
Lee, J. M., Seo, W. Y., Han, H. S., Oh, K. J., Lee, Y. S., Kim, D. K. & Koo, S. H. (2016). Insulin-inducible SMILE inhibits hepatic gluconeogenesis. Diabetes, 65(1), 62-73. Assay: Glycogen in mice liver tissue.
Tarrant, D. J., Stirpe, M., Rowe, M., Howard, M. J., von der Haar, T., & Gourlay, C. W. (2016). Inappropriate expression of the translation elongation factor 1A disrupts genome stability and metabolism. J Cell Sci, 129(24), 4455-4465. Assay: Glycogen in yeast cells.
Poleti, M.D., et al (2015). Genetic variants in glucocorticoid and mineralocorticoid receptors are associated with concentrations of plasma cortisol, muscle glycogen content, and meat quality traits in male Nellore cattle. Domest Anim Endocrinol 51:105-13. Assay: Glycogen in bovine muscle tissue.
Tucci, S., Pearson, S., Herebian, D., & Spiekerkoetter, U. (2013). Long-term dietary effects on substrate selection and muscle fiber type in very-long-chain acyl-CoA dehydrogenase deficient (VLCAD-/-) mice. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 1832(4): 509-516. Assay: Glycogen in mouse skeletal muscle, liver.
Yamamotoya, T., Dose, H., Tian, Z., Faure, A., Toya, Y., Honma, M.,.& Matsuno, H. (2012). Glycogen is the primary source of glucose during the lag phase of E. coli proliferation. Biochimica et Biophysica Acta (BBA)-Proteins and Proteomics 1824(12): 1442-1448. Assay: Glycogen in human buccal, skin, vaginal eluate.
Bouitbir, J., Charles, A. L., Rasseneur, L., Dufour, S., Piquard, F., Geny, B., & Zoll, J. (2011). Atorvastatin treatment reduces exercise capacities in rats: involvement of mitochondrial impairments and oxidative stress. Journal of Applied Physiology 111(5): 1477-1483. Assay: Glycogen in rat muscle tissue.
Iwabuchi S, Kawahara K (2011). Inducible astrocytic glucose transporter-3 contributes to the enhanced storage of intracellular glycogen during reperfusion after ischemia. Neurochem Int 59(2):319-25. Assay: Glycogen in rat astrocytes.
Primassin, S., S. Tucci, et al. (2011). “Hepatic and muscular effects of different dietary fat content in VLCAD deficient mice.” Mol Genet Metab 104(4): 546-51. Assay: Glycogen in mouse muscle, liver.
Yin, M., et al. (2011). Metformin improves cardiac function in a nondiabetic rat model of post-MI heart failure. Am J Physiol Heart Circ Physiol 301(2):H459-68. Assay: Glycogen in rat cardiac muscle.
To find more recent publications, please click here.
If you or your labs do not have the equipment or scientists necessary to run this assay, BioAssay Systems can perform the service for you.
– Fast turnaround
– Quality data
– Low cost
Please email or call 1-510-782-9988 x 2 to discuss your projects.
$499.00
For bulk quote or custom reagents, please email or call 1-510-782-9988 x 1.
Orders are shipped the same day if placed by 2pm PST
Shipping: On Ice
Carrier: Fedex
Delivery: 1-2 days (US), 3-6 days (Intl)
Storage: -20°C upon receipt
Related Products
You may also like…
| Name | SKU | Price | Buy |
|---|---|---|---|
| EnzyChrom™ Starch Assay Kit | E2ST-100 | $479.00 | |
| QuantiChrom™ Glucose Assay Kit | DIGL-100 | $359.00 |
Why BioAssay Systems
Quality and User-friendly • Expert Technical Support • Competitive Prices • Expansive Catalogue • Trusted Globally
