QuantiChrom™ Glutathione (GSH) Assay Kit
Application
- For quantitative determination of reduced glutathione (GSH) and evaluation of drug effects on glutathione metabolism.
Key Features
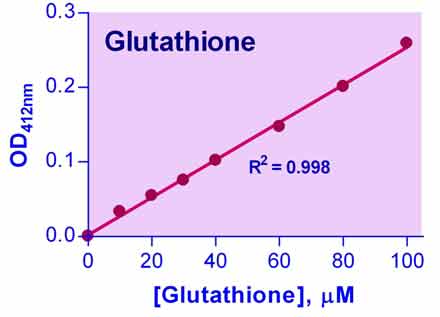
- Sensitive and accurate. Linear detection range 0.4 – 100 µM in 96-well plate.
- Simple and convenient. The procedure involves mixing the DTNB Reagent with sample, removing protein precipitates for proteinaceous samples, adding a second Reagent and reading the optical density.
- Low interference. Amino acids and common buffers do not interfere.
Method
- OD412nm
Samples
- Whole blood, plasma, serum, urine, tissue and cell extracts
Species
- All
Procedure
- 30 min
Size
- 250 tests
Detection Limit
- 12 µg/dL (0.4 µM)
Shelf Life
- 12 months
More Details
Glutathione is a tripeptide of glycine, glutamic acid and cysteine. In the red blood cell, the reduced form of glutathione is vital in maintaining hemoglobin in a reduced state and hence protecting the cells from oxidative damage. Glutathione is involved in detoxification of hydrogen peroxide through glutathione oxidase. Low levels of glutathione are found in deficiencies of key enzymes involved in glutathione metabolism, such as glucose-6-phosphate dehydrogenase, glutathione synthase and glutathione reductase. Simple, direct and automation-ready procedures for measuring reduced glutathione are becoming popular in Research and Drug Discovery. BioAssay Systems QuantiChrom™ Glutathione Assay Kit is designed to accurately measure reduced glutathione in biological samples. The improved 5,5’-dithiobis(2-nitrobenzoic acid (DTNB) method combines deproteination and detection (Reagent A) into one reagent. DTNB reacts with reduced glutathione to form a yellow product. The optical density, measured at 412 nm, is directly proportional to glutathione concentration in the sample. The optimized formulation has a long shelf life and is completely free of interference due to sample turbidity.We would like to know if the QuantiChrom glutathione assay kit ref: DIGT-250 can be used to detect glutathione in wine.
We tested a few wine samples (red and white) with our DIGT-250 kit. Unfortunately, the wine gave too high of a background at 412 nm and the glutathione levels are too low in wine for dilution of the sample to work. Therefore, we cannot recommend using this kit for glutathione determination in wine.
The protocol does not mention sample collection methods (i.e. whether it requires special preservatives, stabilizers etc.). What are your recommendations?
We recommend processing serum samples immediately and avoiding prolonged storage. Rapid acidification is required to prevent GSH auto-oxidation, GSH and GSSG degradation, as well as to precipitate proteins that interfere with analysis. Although we have not validated it, we believe the processed samples should be stable at -80°C.
Hsieh, P.-S., et al. (2021). Lactobacillus spp. Reduces ethanol-induced liver oxidative stress and inflammation in a mouse model of alcoholic steatohepatitis. Experimental and Therapeutic Medicine, 21(3), 188. Assay: Glutathione in mouse tissue and blood.
AlAsmari, A. F., et al. (2020). Elucidation of the molecular mechanisms underlying sorafenib-induced hepatotoxicity. Oxidative Medicine and Cellular Longevity, 2020, 7453406. Assay: Glutathione in rat liver tissue.
Humam, A. M., et al. (2020). Supplementation of postbiotic RI11 improves antioxidant enzyme activity, upregulated gut barrier genes, and reduced cytokine, acute phase protein, and heat shock protein 70 gene expression levels in heat-stressed broilers. Poultry Science, 100908. Assay: Glutathione in chicken plasma.
Kim, N. Y., et al. (2020). Anti-inflammatory effects of ribes diacanthum pall mediated via regulation of nrf2/ho-1 and nf-kb signaling pathways in lps-stimulated raw 264. 7 macrophages and a tpa-induced dermatitis animal model. Antioxidants (Basel, Switzerland), 9(7). Assay: Glutathione in mouse macrophage cells.
Sahin, T. D., et al. (2020). Resveratrol and quercetin attenuate depressive-like behavior and restore impaired contractility of vas deferens in chronic stress-exposed rats: Involvement of oxidative stress and inflammation. Naunyn-Schmiedeberg ‘s Archives of Pharmacology, 393(5), 761-775. Assay: Glutathione in rat tissue.
Sahin, T. D., et al. (2020). Infliximab prevents dysfunction of the vas deferens by suppressing inflammation and oxidative stress in rats with chronic stress. Life Sciences, 250, 117545. Assay: Glutathione in rat tissue.
Humam, A. M., et al (2020). Dietary supplementation of postbiotics mitigates adverse impacts of heat stress on antioxidant enzyme activity, total antioxidant, lipid peroxidation, physiological stress indicators, lipid profile and meat quality in broilers. Animals: An Open Access Journal from MDPI, 10(6). Assay: Glutathione in chicken plasma.
Pengrattanachot, N., et al (2020). Atorvastatin attenuates obese-induced kidney injury and impaired renal organic anion transporter 3 function through inhibition of oxidative stress and inflammation. Biochimica Et Biophysica Acta. Molecular Basis of Disease, 1866(6), 165741. Assay: Glutathione in rat renal tissue.
Shin, S. K., et al. (2019). Mulberry fruit improves memory in scopolamine-treated mice: Role of cholinergic function, antioxidant system, and TrkB/Akt signaling. Nutritional Neuroscience, 1-11. Assay: Glutathione in mouse tissue.
Sowmithra, S., et al (2020). Recovery of human embryonic stem cells-derived neural progenitors exposed to hypoxic-ischemic-reperfusion injury by indirect exposure to wharton ‘s jelly mesenchymal stem cells through phosphatidyl-inositol-3-kinase pathway. Cellular and Molecular Neurobiology. Assay: Glutathione in human cells.
Lee, B. D., et al (2019). 3,3 ‘-diindolylmethane promotes bdnf and antioxidant enzyme formation via trkb/akt pathway activation for neuroprotection against oxidative stress-induced apoptosis in hippocampal neuronal cells. Antioxidants (Basel, Switzerland), 9(1). Assay: Glutathione in mouse cells and tissue.
Liang, H., et al (2020). N-acetyl serotonin alleviates oxidative damage by activating nuclear factor erythroid 2-related factor 2 signaling in porcine enterocytes. Antioxidants, 9(4), 303. Assay: Glutathione in pig cells.
Pena-Bermudez, Y. A., et al (2020). Effects of feeding increasing levels of yerba mate on lamb meat quality and antioxidant activity. Animals: An Open Access Journal from MDPI, 10(9). Assay: Glutathione in sheep tissue.
Min, A. Y., et al (2020). Mulberry fruit prevents diabetes and diabetic dementia by regulation of blood glucose through upregulation of antioxidative activities and creb/bdnf pathway in alloxan-induced diabetic mice. Oxidative Medicine and Cellular Longevity, 2020, 1298691. Assay: Glutathione in mouse organ tissue.
Hwang, J., et al (2020). Genome sequence of the potential probiotic eukaryote Saccharomyces cerevisiae KCCM 51299. 3 Biotech, 10(4), 185. Assay: Glutathione in yeast media.
Hassannia, B., Wiernicki, B., Ingold, I., Qu, F., Van Herck, S., Tyurina, Y. Y. & Meul, E. (2018). Nano-targeted induction of dual ferroptotic mechanisms eradicates high-risk neuroblastoma. Journal of Clinical Investigation, 128(8), 3341-3355. Assay: Glutathione in human cells.
Lee, H. J., Han, J. H., Park, Y. K., & Kang, M. H. (2018). Effects of glutathione s-transferase (GST) M1 and T1 polymorphisms on antioxidant vitamins and oxidative stress-related parameters in Korean subclinical hypertensive subjects after kale juice (Brassica oleracea acephala) supplementation. Nutrition research and practice, 12(2), 118-128. Assay: Glutathione in human blood.
Mizrahi, M., Adar, T., Lalazar, G., Nachman, D., El Haj, M., Ya’acov, A. B. & Ilan, Y. (2018). Glycosphingolipids Prevent APAP and HMG-CoA Reductase Inhibitors-mediated Liver Damage: A Novel Method for “Safer Drug” Formulation that Prevents Drug-induced Liver Injury. Journal of clinical and translational hepatology 6(2): 127-134. Assay: Glutathione in mice cells.
Saint-Germain, E., Mignacca, L., Vernier, M., Bobbala, D., Ilangumaran, S., & Ferbeyre, G. (2017). SOCS1 regulates senescence and ferroptosis by modulating the expression of p53 target genes. Aging (Albany NY) 9(10): 2137-2162. Assay: Glutathione in human cells.
Jaikumkao, K., Pongchaidecha, A., Chattipakorn, N., Chatsudthipong, V., Promsan, S., Arjinajarn, P., & Lungkaphin, A. (2016). Atorvastatin improves renal organic anion transporter 3 and renal function in gentamicin-induced nephrotoxicity in rats. Experimental physiology, 101(6), 743-753. Assay: Glutathione in Sprague Dewley rats renal tissue.
Suh, KS et al (2016). Protective effects of honokiol against methylglyoxal-inducedosteoblast damage. Chemico-Biological Interactions. 244:169-177. Assay: Glutathione in plant extract cells.
Barros, MA et al (2015). L-Alanyl-Glutamine Attenuates Oxidative Stress in Liver Transplantation Patients. Transplantation Proceedings. 47(8):2478-82. Assay: Glutathione in human liver tissue.
Malek, HA et al (2015). The preventive effect of beta3 adrenoceptor stimulation against experimentally induced reflux esophagitis. Acta Physiologica Hungarica. 102(1): 94-104. Assay: Glutathione in rat plasma.
Mesci, P et al (2015). System xC- is a mediator of microglial function and its deletion slows symptoms in amyotrophic lateral sclerosis mice. Brain. 138(Pt 1):53-68. Assay: Glutathione in mice tissue.
Yun, J et al (2015). Bergenin decreases the morphine-induced physical dependence via antioxidative activity in mice. Archives of Pharmacal Research. 38(6):1248-1254. Assay: Glutathione in mice brain and liver tissue.
Kaur C, et al (2010). Melatonin protects periventricular white matter from damage due to hypoxia. J Pineal Res. 48(3):185-93. Assay: Glutathione in rat brain tissue.
Labib, HM, et al (2010). The Role of Oxidative Stress Markers and Nitric Oxide Levels in the Pathogenesis of Glaucoma. Austr. J. Basic and Applied Sci 4(8): 3553-3558. Assay: Glutathione in human blood.
Ogunrinu TA, Sontheimer H (2010). Hypoxia increases the dependence of glioma cells on glutathione. J Biol Chem. 285(48):37716-24. Assay: Glutathione in human glioma cells.
Park, MS et al (2010). Korean Red Ginseng Protects Oxidative Injury Caused by Lead Poisoning. JGR 34(2):132-137. Assay: Glutathione in rat blood.
Park, MS et al (2010). Korean Red Ginseng Protects Oxidative Injury Caused by Lead Poisoning. JGR 34(2):132-137. Assay: Glutathione in rat blood.
Du Y, Villeneuve NF, Wang XJ, Sun Z, Chen W, Li J, Lou H, Wong PK, Zhang DD (2008). Oridonin confers protection against arsenic-induced toxicity through activation of the Nrf2-mediated defensive response. Environ Health Perspect.116(9):1154-61. Assay: Glutathione in human cell lines.
Gumpricht E, et al (2008). Resistance of young rat hepatic mitochondria to bile acid-induced permeability transition: potential role of alpha-tocopherol. Pediatr Res. 64(5):498-504. Assay: Glutathione in rat mitochondrial GSH.
To find more recent publications, please click here.
If you or your labs do not have the equipment or scientists necessary to run this assay, BioAssay Systems can perform the service for you.
– Fast turnaround
– Quality data
– Low cost
Please email or call 1-510-782-9988 x 2 to discuss your projects.
$499.00
For bulk quote or custom reagents, please email or call 1-510-782-9988 x 1.
Orders are shipped the same day if placed by 2pm PST
Shipping: RT
Carrier: Fedex
Delivery: 1-2 days (US), 3-6 days (Intl)
Storage: 4°C upon receipt
Related Products
You may also like…
| Name | SKU | Price | Buy |
|---|---|---|---|
| QuantiChrom™ Glutathione S-transferase Assay Kit | DGST-100 |
$389.00 |
Why BioAssay Systems
Quality and User-friendly • Expert Technical Support • Competitive Prices • Expansive Catalogue • Trusted Globally
