QuantiChrom™ BCP Albumin Assay Kit
Application
- For quantitative determination of albumin and evaluation of drug effects on albumin metabolism.
Key Features
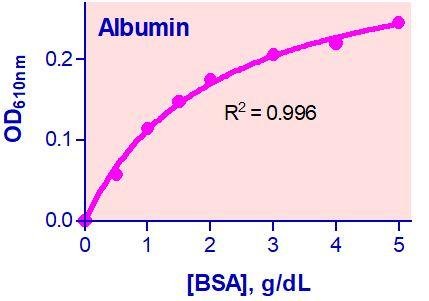
- Sensitive and accurate. Use as little as 20 µL samples. Detection range 0.3 – 5 g/dL (45 – 750 µM) albumin in a 96-well plate assay.
- Simple and high-throughput. The procedure involves the addition of a single working reagent and incubation for 5 min. Can be readily automated as a high-throughput assay for thousands of samples per day.
- Improved reagent stability and versatility. The optimized formulation has greatly enhanced the reagent and signal stability. Cuvet or 96-well plate assay.
- Low interference in biological samples. No pretreatments are needed. Assays can be directly performed on raw biological samples i.e., in the presence of lipids and protein.
Method
- OD610nm (BCP)
Samples
- Serum, plasma, urine, biological preparations
Species
- All
Procedure
- 5 min
Size
- 250 tests
Detection Limit
- 0.3 g/dL
Shelf Life
- 12 months
More Details
Albumin is the most abundant plasma protein in humans. It accounts for about 60% of the total serum protein. Albumin plays important physiological roles, including the maintenance of colloid osmotic pressure, and binding of key substances such as long-chain fatty acids, bile acids, bilirubin, haematin, calcium, and magnesium. It has anti-oxidant and anticoagulant effects, and also acts as a carrier for nutritional factors and drugs, as an effective plasma pH buffer. Serum albumin is a reliable prognostic indicator for morbidity and mortality, liver disease, nephritic syndrome, malnutrition, and protein-losing enteropathies. High levels are associated with dehydration. Simple, direct, and automation-ready procedures for measuring albumin concentration in biological samples are becoming popular in Research and Drug Discovery. BioAssay Systems BCP albumin assay kit is designed to measure albumin directly in biological samples without any pretreatment. The improved method utilizes bromcresol purple that forms a colored complex specifically with albumin. The intensity of the color, measured at 610nm, is directly proportional to the albumin concentration in the sample. The optimized formulation substantially reduces interference by substances in the raw samples.Your company sells two different kit (DIAG-250 and DIAP-250). What are the differences between them exactly? Is it just the wavelength of detection?
The two kits use different albumin specific dyes and hence the major difference lies in the sensitivity and wavelengths. Both assays have advantages and disadvantages. DIAG-250 (QuantiChrom BCG Albumin Assay Kit) uses bromcresol green, a dye that is more (about 2-fold) sensitive than bromcresol purple used in the BCP albumin assay. BCG is not absolutely specific for albumin, however, it also reacts with α1- and α2-globulins, and adds a positive bias. The BCP method, although more specific, underestimates albumin in certain samples, e.g. from patients undergoing hemodialysis. If your samples are sera or plasma, which have high albumin concentrations, I would suggest using the DIAP-250 assay kit (detection wavelength 610 nm). If the albumin concentration is low, DIAG-250 is the best choice (detection wavelength 620 nm).
Pooled plasma samples (EDTA) show high albumin concentrations with the bromcresol purple assay? What could be interfering with the assay?
There should be no significant differences between serum samples and plasma samples. Turbid plasma samples (fibrinogen) can cause high albumin readings. This can be adjusted for by blanking samples at OD 690 nm. BCP does not absorb at 690 nm, whereas the absorbance caused by turbididty is the same at 610 nm and 690 nm.
Since the concentration that I am measuring is very low, would it be reasonable to dilute the standard curve down to lower level so my sample is still within the curve?
Yes, as long as the standard curve is within the limits of the dynamic range, but this will not increase the sensitivity of the assay. To increase the sensitivity add a larger volume of sample and standard. For example, you could add 20 μL sample and 20 μL of diluted standards (instead of starting at 5 g/dL as shown in the standard protocol, start at 1 g/dL).
Ben Othman, S., et al. (2020). Senescence-accelerated mouse prone 8 mice exhibit specific morphological changes in the small intestine during senescence and after pectin supplemented diet. Experimental Gerontology 142. Assay: Albumin in mouse serum.
Kelly E, et al (2011). Redistribution of labile plasma zinc during mild surgical stress in the rat. Transl Res. 157(3):139-49. Assay: Albumin in rat plasma.
Olsen AS, et al (2010). Limb regeneration is impaired in an adult zebrafish model of diabetes mellitus. Wound Repair Regen. 18(5):532-42. Assay: Albumin in zebrafish blood.
Sharifuzzaman SM and Austin B (2010). Kocuria SM1 controls vibriosis in rainbow trout (Oncorhynchus mykiss, Walbaum). J Appl Microbiol. 108(6): 2162 – 2170. Assay: Albumin in fish serum.
Shin SY et al (2009). Immunological investigation in the adenoid tissues from children with chronic rhinosinusitis. Otolaryngol Head Neck Surg. 141(1):91-6. Assay: Albumin in human tissue.
Shin SY, et al (2009). IgE response to staphylococcal enterotoxins in adenoid tissues from atopic children. Laryngoscope. 119(1):171-5. Assay: Albumin in human serum.
Shin SY, et al (2009). Local production of total IgE and specific antibodies to the house dust mite in adenoid tissue. Pediatr Allergy Immunol. 20(2):134-41. Assay: Albumin in human blood.
Yung S, et al (2009). Anti-DNA antibody induction of protein kinase C phosphorylation and fibronectin synthesis in human and murine lupus and the effect of mycophenolic acid. Arthritis Rheum. 60(7):2071-82. Assay: Albumin in human urine.
Maier SM et al (2007) Proteinuria of Nonautoimmune Origin in Wild-type FVB/NJ Mice. Comp Med. 57(3) 255-266. Assay: Albumin in mouse urine and serum.
To find more recent publications, please click here.
If you or your labs do not have the equipment or scientists necessary to run this assay, BioAssay Systems can perform the service for you.
– Fast turnaround
– Quality data
– Low cost
Please email or call 1-510-782-9988 x 2 to discuss your projects.
$419.00
For bulk quote or custom reagents, please email or call 1-510-782-9988 x 1.
Orders are shipped the same day if placed by 2pm PST
Shipping: RT
Carrier: Fedex
Delivery: 1-2 days (US), 3-6 days (Intl)
Storage: -20°C upon receipt
Related Products
You may also like…
| Name | SKU | Price | Buy |
|---|---|---|---|
| QuantiChrom™ BCG Albumin Assay Kit | DIAG-250 | $429.00 | |
| QuantiChrom™ Hemoglobin Assay Kit | DIHB-250 | $449.00 |
Why BioAssay Systems
Quality and User-friendly • Expert Technical Support • Competitive Prices • Expansive Catalogue • Trusted Globally
