QuantiChrom™ BCG Albumin Assay Kit
Application
- For quantitative determination of albumin and evaluation of drug effects on albumin metabolism.
Key Features
- Sensitive and accurate. Use as little as 5 µL samples. Detection range 0.01 – 5 g/dL (1.5 – 750 µM) albumin in a 96-well plate assay.
- Simple and high-throughput. The procedure involves the addition of a single working reagent and incubation for 5 min. Can be readily automated as a high-throughput assay in 96-well plates for thousands of samples per day.
- Improved reagent stability and versatility. The optimized formulation has greatly enhanced the reagent and signal stability. Cuvet or 96-well plate assay.
- No interference in biological samples. No pretreatments are needed. Assays can be directly performed on raw biological samples i.e., in the presence of lipids and proteins.
Method
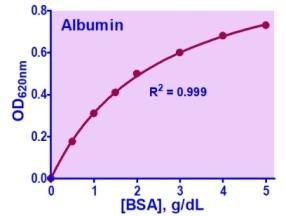
- OD620nm (BCG)
Samples
- Serum, plasma, urine, biological preparations
Species
- All
Procedure
- 5 min
Size
- 250 tests
Detection Limit
- 0.01 g/dL
Shelf Life
- 12 months
More Details
Albumin is the most abundant plasma protein in humans. It accounts for about 60% of the total serum protein. Albumin plays important physiological roles, including the maintenance of colloid osmotic pressure, and binding of key substances such as long-chain fatty acids, bile acids, bilirubin, haematin, calcium, and magnesium. It has anti-oxidant and anticoagulant effects, and also acts as a carrier for nutritional factors and drugs, as an effective plasma pH buffer. Serum albumin is a reliable prognostic indicator for morbidity and mortality, liver disease, nephritic syndrome, malnutrition, and protein-losing enteropathies. High levels are associated with dehydration. Simple, direct, and automation-ready procedures for measuring albumin concentration in biological samples are becoming popular in Research and Drug Discovery. BioAssay Systems BCG albumin assay kit is designed to measure albumin directly in biological samples without any pretreatment. The improved method utilizes bromcresol green that forms a colored complex specifically with albumin. The intensity of the color, measured at 620nm, is directly proportional to the albumin concentration in the sample. The optimized formulation substantially reduces interference by substances in the raw samples.Your company sells two different kit (DIAG-250 and DIAP-250). What are the differences between them exactly? Is it just the wavelength of detection?
The two kits use different albumin specific dyes and hence the major difference lies in the sensitivity and wavelengths. Both assays have advantages and disadvantages. DIAG-250 (QuantiChrom BCG Albumin Assay Kit) uses bromcresol green, a dye that is more (about 2-fold) sensitive than bromcresol purple used in the BCP albumin assay. BCG is not absolutely specific for albumin, however, it also reacts with α1- and α2-globulins, and adds a positive bias. The BCP method, although more specific, underestimates albumin in certain samples, e.g. from patients undergoing hemodialysis. If your samples are sera or plasma, which have high albumin concentrations, I would suggest using the DIAP-250 assay kit (detection wavelength 610 nm). If the albumin concentration is low, DIAG-250 is the best choice (detection wavelength 620 nm).
Pooled plasma samples (EDTA) show high albumin concentrations with the bromcresol purple assay? What could be interfering with the assay?
There should be no significant differences between serum samples and plasma samples. Turbid plasma samples (fibrinogen) can cause high albumin readings. This can be adjusted for by blanking samples at OD 690 nm. BCP does not absorb at 690 nm, whereas the absorbance caused by turbididty is the same at 610 nm and 690 nm.
Since the concentration that I am measuring is very low, would it be reasonable to dilute the standard curve down to lower level so my sample is still within the curve?
Yes, as long as the standard curve is within the limits of the dynamic range, but this will not increase the sensitivity of the assay. To increase the sensitivity add a larger volume of sample and standard. For example, you could add 20 μL sample and 20 μL of diluted standards (instead of starting at 5 g/dL as shown in the standard protocol, start at 1 g/dL).
Zhao, C., et al(2020). Cyclin G2 regulates canonical Wnt signalling via interaction with Dapper1 to attenuate tubulointerstitial fibrosis in diabetic nephropathy. Journal of Cellular and Molecular Medicine, 24(5), 2749-2760. Assay: Albumin in Mice Urinary Albumin.
Xu, D., et al (2018). NIX-mediated mitophagy protects against proteinuria-induced tubular cell apoptosis and renal injury. American Journal of Physiology-Renal Physiology, 316(2) Assay: Albumin in Mice Urinary Albumin.
Manna, L., et al (2020). Impact of Phyllantus niruri and Lactobacillus amylovorus SGL 14 in a mouse model of dietary hyperoxaluria. Beneficial Microbes, 11(6), 547-559. Assay: Albumin in Mice Kidney Tissue.
Sethi, P., et al (2019). Plasma protein and lipoprotein binding of cis- and trans-permethrin and deltamethrin in adult humans and rats. Drug Metabolism and Disposition: The Biological Fate of Chemicals, 47(9), 941-948. Assay: Albumin in Rat and Human Plasma.
Park, S.-J., et al (2019). Discovery of endoplasmic reticulum calcium stabilizers to rescue ER-stressed podocytes in nephrotic syndrome. Proceedings of the National Academy of Sciences, 116(28), 14154-14163. Assay: Albumin in Mice Urine.
Amor, C., et al (2020). Senolytic CAR T cells reverse senescence-associated pathologies. Nature, 583(7814), 127-132. Assay: Albumin in Murine Serum.
Parchem, J. G., et al (2021). STOX1 deficiency is associated with renin-mediated gestational hypertension and placental defects. JCI Insight, 6(2). Assay: Albumin in Mice Urine Albumin.
Liu, X., et al (2019). Metabolomics analysis reveals the protection mechanism of huangqi-danshen decoction on adenine-induced chronic kidney disease in rats. Frontiers in Pharmacology, 10. Assay: Albumin in Male Sprague-Dawley Rats Urine.
Blom, E. (2019). Receiving and backgrounding phase management: The effects of receiving diet roughage source and backgrounding phase rate of gain on performance of feedlot cattle. Electronic Theses and Dissertations. Assay: Albumin in Angus-based crossbred steer calves Blood.
Proffitt, S., Curnow, E., Brown, C., Bashir, S., & Cardigan, R. (2018). Comparison of automated and manual methods for washing red blood cells. Transfusion, 58(9), 2208-2216. Assay: Albumin in red blood cells.
Rangel, E. B., Gomes, S. A., Kanashiro-Takeuchi, R., Saltzman, R. G., Wei, C., Ruiz, P. & Hare, J. M. (2018). Kidney-derived c-kit+ progenitor/stem cells contribute to podocyte recovery in a model of acute proteinuria. Scientific reports, 8(1), 14723. Assay: Albumin in rats blood.
Shepardson, K. M., Larson, K., Johns, L. L., Stanek, K., Cho, H., Wellham, J. & Rynda-Apple, A. (2018). IFNAR2 is required for anti-influenza immunity and alters susceptibility to post-influenza bacterial superinfections. Frontiers in Immunology 9:2589. Assay: Albumin in mice cell free BAL fluid.
Xu, D. et al(2018). NIX-mediated mitophagy protects against proteinuria-induced tubular cell apoptosis and renal injury. American Journal of Physiology-Renal Physiology, 316(2), F382-F395. Assay: Albumin in mice epithelial cells.
Chuang, M. H., Jan, M. S., Chang, J. T., & Lu, F. J. (2017). The Chinese medicine JC-001 enhances the chemosensitivity of Lewis lung tumors to cisplatin by modulating the immune response. BMC complementary and alternative medicine, 17(1), 210. Assay: Albumin in human venous blood.
Garnier, R., Bento, A. I., Hansen, C., Pilkington, J. G., Pemberton, J. M., & Graham, A. L. (2017). Physiological proteins in resource-limited herbivores experiencing a population die-off. The Science of Nature, 104(7-8), 68. Assay: Albumin in sheep plasmatic albumin.
Garnier, R., Cheung, C. K., Watt, K. A., Pilkington, J. G., Pemberton, J. M., & Graham, A. L. (2017). Joint associations of blood plasma proteins with overwinter survival of a large mammal. Ecology letters, 20(2), 175-183. Assay: Albumin in sheep plasmatic albumin.
Hassanein, W. et al (2017). Liver Scaffolds Support Survival and Metabolic Function of Multilineage Neonatal Allogenic Cells. Tissue Engineering Part A, 24(9-10), 786-793. Assay: Albumin in rats liver cells.
Hassanein, W., Uluer, M. C., Langford, J., Woodall, J. D., Cimeno, A., Dhru, U. & Khalifeh, A. (2017). Recellularization via the bile duct supports functional allogenic and xenogenic cell growth on a decellularized rat liver scaffold. Organogenesis, 13(1), 16-27. Assay: Albumin in human liver cells.
Kingman, J., Uitto, J., & Li, Q. (2017). Elevated dietary magnesium during pregnancy and postnatal life prevents ectopic mineralization in Enpp1asj mice, a model for generalized arterial calcification of infancy. Oncotarget, 8(24), 38152. Assay: Albumin in mice urinary albumin.
Van Le, T., Nguyen, N. H., Do, H. Q., Le, H. M., & Truong, N. H. (2017). Transplantation of umbilical cord blood-derived mesenchymal stem cells to treat liver cirrhosis in mice: a comparison of tail and portal vein injection. Progress in Stem Cell, 4(2), 201-216. Assay: Albumin in human liver cells.
Blair, T. C., Manoharan, M., Rawlings-Rhea, S. D., Tagge, I., Kohama, S. G., Hollister-Smith, J. & Rooney, W. D. (2016). Immunopathology of Japanese macaque encephalomyelitis is similar to multiple sclerosis. Journal of neuroimmunology, 291, 1-10. Assay: Albumin in macaque .
Frej, C., Linder, A., Happonen, K. E., Taylor, F. B., Lupu, F., & Dahlback, B. (2016). Sphingosine 1-phosphate and its carrier apolipoprotein M in human sepsis and in Escherichia coli sepsis in baboons. Journal of cellular and molecular medicine, 20(6), 1170-1181. Assay: Albumin in human, baboon albumin.
Fujino, T., & Hasebe, N. (2016). Alteration of histone H3K4 methylation in glomerular podocytes associated with proteinuria in patients with membranous nephropathy. BMC nephrology, 17(1), 179. Assay: Albumin in mouse cells.
Lei, G. S., Zhang, C., Zimmerman, M. K., & Lee, C. H. (2016). Vitamin D as supplemental therapy for Pneumocystis pneumonia. Antimicrobial agents and chemotherapy, 60(3), 1289-1297. Assay: Albumin in mice cell free BAL fluid.
Sethi, P. K., White, C. A., Cummings, B. S., Hines, R. N., Muralidhara, S., & Bruckner, J. V. (2016). Ontogeny of plasma proteins, albumin and binding of diazepam, cyclosporine, and deltamethrin. Pediatric research, 79(3), 409. Assay: Albumin in human plasmatic albumin.
Truong, N. H., Nguyen, N. H., Le, T. V., Vu, N. B., Huynh, N., Nguyen, T. V. & Pham, P. V. (2016). Comparison of the treatment efficiency of bone marrow-derived mesenchymal stem cell transplantation via tail and portal veins in CCl4-induced mouse liver fibrosis. Stem Cells International, 2016. Assay: Albumin in mice venous blood.
Tsang, G., Insel, M. B., Weis, J. M., Morgan, M. A. M., Gough, M. S., Frasier, L. M. & Pietropaoli, A. P. (2016). Bioavailable estradiol concentrations are elevated and predict mortality in septic patients: a prospective cohort study. Critical Care, 20(1), 335. Assay: Albumin in human venous blood.
Yokoyama, S., Hiramoto, K., Koyama, M., & Ooi, K. (2016). Chronic liver injury in mice promotes impairment of skin barrier function via tumor necrosis factor-alpha. Cutaneous and ocular toxicology, 35(3), 194-203. Assay: Albumin in mice plasmatic albumin.
Doi M, Sultana Rekha R, Ahmed S, Okada M, Kumar Roy A, El Arifeen S, Ekstrom EC, Raqib R, Wagatsuma Y. (2011). Association between calcium in cord blood and newbornsize in Bangladesh. Br J Nutr. 106(9):1398-407. Assay: Albumin in human blood.
Euhus DM, et al (2011). Tamoxifen Downregulates Ets-oncogene Family Members ETV4 and ETV5 in Benign Breast Tissue: Implications for Durable Risk Reduction. Cancer Prev Res (Phila). 4(11):1852-62. Assay: Albumin in human blood.
Zhang G, et al (2011). Nicotinic acetylcholine receptor alpha1 promotes calpain-1 activation and macrophage inflammation in hypercholesterolemic nephropathy. Lab Invest. 91(1):106-23. Assay: Albumin in mouse urine.
Irani K, et al (2010). Mechanical dissociation of swine liver to produce organoid units for tissue engineering and in vitro disease modeling. Artif Organs. 34(1):75-8. Assay: Albumin in swine hepatocyte.
Roberts DJ, et al (2009). Effect of acute inflammatory brain injury on accumulation of morphine and morphine 3- and 6-glucuronide in the human brain. Crit Care Med. 37(10):2767-74. Assay: Albumin in human plasma and cerebrospinal fluid.
Cosgrove, D. et al (2008). Integrin alpha1beta1 Regulates Matrix Metalloproteinases via P38 Mitogen-Activated Protein Kinase in Mesangial Cells. Implications for Alport Syndrome. Am. J. Pathology 172(3):761-73. Assay: Albumin in mouse urine.
Helsby, NA et al (2008). CYP2C19 pharmacogenetics in advanced cancer: compromised function independent of genotype. Br J Cancer. 99(8):1251-5. Assay: Albumin in human serum.
Kain R, et al (2008). Molecular mimicry in pauci-immune focal necrotizing glomerulonephritis. Nat Med. 14(10):1088-96. Assay: Albumin in human albuminuria.
Osborn TM, et al (2008). Decreased levels of the gelsolin plasma isoform in patients with rheumatoid arthritis. Arthritis Res Ther. 10(5):R117. Assay: Albumin in human plasma and synovial fluid.
Sullivan JP, et al (2008). Convection and hemoglobin-based oxygen carrier enhanced oxygen transport in a hepatic hollow fiber bioreactor. Artif Cells Blood Substit Immobil Biotechnol.36(4):386-402. Assay: Albumin in human hepatocyte.
Lee, R.H. et al (2006) Multipotent stromal cells from human marrow home to and promote repair of pancreatic islets and renal glomeruli in diabetic NOD_scid mice. PNAS 103 (46): 17438-17443. Assay: Albumin in mouse urine.
Rebecca R. (2006). Associations of histories of depression and PMDD diagnosis with allopregnanolone concentrations following the oral administration of micronized progesterone sychoneuroendocrinology 31(10):1208-1219. Assay: Albumin in human serum.
Wang S, et al (2006). Renal bone morphogenetic protein-7 protects against diabetic nephropathy. J Am Soc Nephrol. 17(9):2504-12. Assay: Albumin in mouse serum.
Zhang Q, et al (2006). Serum and mucosal antibody responses to pneumococcal protein antigens in children: relationships with carriage status. Eur J Immunol.36(1):46-57. Assay: Albumin in human saliva and serum.
To find more recent publications, please click here.
If you or your labs do not have the equipment or scientists necessary to run this assay, BioAssay Systems can perform the service for you.
– Fast turnaround
– Quality data
– Low cost
Please email or call 1-510-782-9988 x 2 to discuss your projects.
$429.00
For bulk quote or custom reagents, please email or call 1-510-782-9988 x 1.
Orders are shipped the same day if placed by 2pm PST
Shipping: RT
Carrier: Fedex
Delivery: 1-2 days (US), 3-6 days (Intl)
Storage: -20°C upon receipt
Related Products
You may also like…
| Name | SKU | Price | Buy |
|---|---|---|---|
| QuantiChrom™ BCP Albumin Assay Kit | DIAP-250 |
$419.00 |
|
| QuantiChrom™ Hemoglobin Assay Kit | DIHB-250 |
$449.00 |
Why BioAssay Systems
Quality and User-friendly • Expert Technical Support • Competitive Prices • Expansive Catalogue • Trusted Globally
