Shake Flask Solubility Service
Summary
The solubility of a drug compound plays a significant role in absorption and is an important factor to consider during development. Quantitative measurement of compound solubility is an ideal early test to perform when screening for potential drug candidates.
BioAssay Systems (BAS) offers two methods for quantifying the solubility of drug compounds: filter plate solubility testing and shake flask solubility testing. This information page details the Shake Flask Method.
BioAssay Systems (BAS) offers two methods for quantifying the solubility of drug compounds: filter plate solubility testing and shake flask solubility testing. This information page details the Shake Flask Method.
Method
In 2 mL tubes, an excess of the test compound is weighed out and mixed with a solvent of customer’s choice. The tubes are shaken for 18 hours (or desired duration) at ambient temperature (or desired temperature). The solution is vacuum filtered to remove precipitates. Absorbance spectrum is run on the filtrate together with the test compound standards. Concentration of the filtrate is calculated using the slope of the standards.
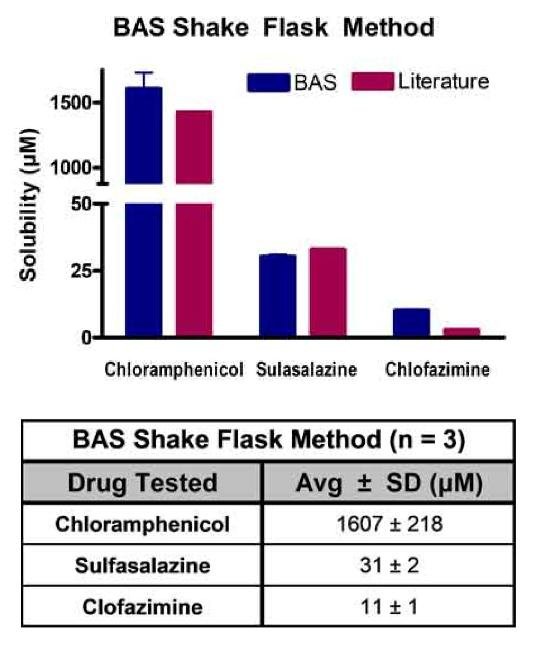
Figure 1: Drug compound solubility determined using the shake flask method. Testing was repeated three times on separate days. Experimentally determined solubility of drug compounds using the shake flask method were compared to literature values.
References
1. Wishart, DS. et al (2018) Nucleic Acids Res. 46(D1): D608-17.
2. PubChem Compound Database; CID=5359476, https://pubchem.ncbi.nlm.nih.gov/compound/5359476.
References
1. Wishart, DS. et al (2018) Nucleic Acids Res. 46(D1): D608-17.
2. PubChem Compound Database; CID=5359476, https://pubchem.ncbi.nlm.nih.gov/compound/5359476.
Our Services
- As little as 50 to 1000 µg compound is needed.
- No structure information is required.
- If compound is in DMSO, 10mM or higher concentration is preferred.
- Report will be available within 72 hours.
Please email or call us at 1-510-782-9988 x 2 to discuss your service needs.