EnzyChrom™ Creatine Assay Kit
Application
- For quantitative determination of creatine and evaluation of drug effects on creatine metabolism.
Key Features
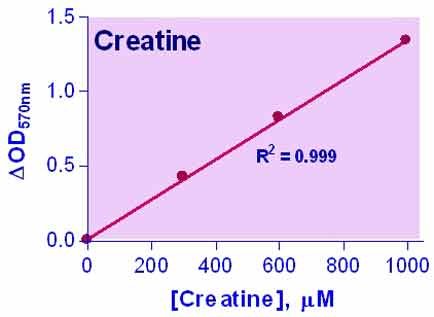
- High sensitivity and wide linear range. Use 10 µL sample. Linear detection range 4 to 1000 µM (colorimetric) or 0.5 to 50 µM (fluorimetric).
- Homogeneous and simple procedure. A simple “mix-and-measure” procedure allows reliable quantitation of creatine within 30 minutes.
Method
- OD570nm, or FL530/590nm
Samples
- Serum, plasma, urine, saliva, etc
Species
- All
Procedure
- 30 min
Size
- 100 tests
Detection Limit
- 4 µM
Shelf Life
- 12 months
More Details
CREATINE is present in vertebrates and helps to supply energy to muscles. In humans and animals, approximately half of creatine originates from food (mainly from fresh meat). Creatine supplementation has been investigated as a possible therapeutic approach for the treatment of muscular, neuromuscular, neurological, and neurodegenerative diseases. Simple, direct, and automation-ready procedures for measuring creatine are popular in research and drug discovery. BioAssay Systems creatine assay is based on enzymatic reactions leading to the formation of a pink-colored product. The optical density at 570 nm or fluorescence intensity at λ ex/em = 530/585 nm is directly proportional to the creatine concentration in the sample.Can this kit be used to measure creatine concentrations in cells?
Yes. To determine intracellular creatine concentrations, harvest cells (0.1-2 million cells per assay) in a 1.5mL tube, centrifuge 1-2 min at 3,000 rpm on a table centrifuge. Remove culture medium, wash cells quickly with cold PBS. Immediately remove any PBS. Prepare Working Reagent. Add 120 µL WR directly to the cell pellet and 80 µL WR to standards in 96-wells. Vortex cell sample tube 1 min, incubate 30 min at room temp. Centrifuge 5 min at 14,000 rpm. Transfer 100 µL supernatant to 96-well. Read OD570nm on a plate reader.
Notes: 1. cell number. It is prudent to run several doses of cells, e.g. 0.1, 1, 2×106 cells to determine optimal cell number to be used in subsequent assays.
Working Reagent should contain a lysis reagent. BioAssay Systems will supply this reagent upon request.
Wang, Yujie, et al (2018). Guanidinoacetic Acid Regulates Myogenic Differentiation and Muscle Growth Through miR-133a-3p and miR-1a-3p Co-mediated Akt/mTOR/S6K Signaling Pathway. International journal of molecular sciences 19.9: 2837. Assay: Creatine in murine cells.
Chan, Yiumo Michael, et al (2017). Substantial deficiency of free sialic acid in muscles of patients with GNE myopathy and in a mouse model. PloS one 12.3: e0173261. Assay: Creatine in human and mouse tissues.
Holz, Josefin-Beate, and Alex Hemeryck (2017). Inhibition of bone resorption with rankl binding peptides. U.S. Patent Application No. 15/295,229. Assay: Creatine in human urine.
Schoborg, JA et al (2014). Substrate replenishment and byproduct removal improve yeast cell-free protein synthesis. Biotechnology Journal 9(5): 630-640. Assay: Creatine in yeast cells.
Qi S et al (2012). Comparison of the metabolic profiling of hepatitis B virus-infected cirrhosis and alcoholic cirrhosis patients by using (1) H NMR-based metabonomics. Hepatol Res 42(7):677-85. Assay: Creatine in human serum.
To find more recent publications, please click here.
If you or your labs do not have the equipment or scientists necessary to run this assay, BioAssay Systems can perform the service for you.
– Fast turnaround
– Quality data
– Low cost
Please email or call 1-510-782-9988 x 2 to discuss your projects.
$499.00
For bulk quote or custom reagents, please email or call 1-510-782-9988 x 1.
Orders are shipped the same day if placed by 2pm PST
Shipping: On Ice
Carrier: Fedex
Delivery: 1-2 days (US), 3-6 days (Intl)
Storage: -20°C upon receipt
Related Products
You may also like…
| Name | SKU | Price | Buy |
|---|---|---|---|
| EnzyChrom™ Creatine Kinase Assay Kit | ECPK-100 |
$599.00 |
|
| QuantiChrom™ Creatinine Assay Kit | DICT-500 |
$479.00 |
Why BioAssay Systems
Quality and User-friendly • Expert Technical Support • Competitive Prices • Expansive Catalogue • Trusted Globally
